Scientists have been trying to find ways of eliminating CO2 to stop global warming, but the practical and economic limitations prevented progress.
However, researchers recently announced a breakthrough that could help save the future of our environment.
At the Goldschmidt conference in Boston, researchers said they found a way to rapidly and cost-effectively create the mineral magnesite in a lab.
If the carbon dioxide-storing mineral can be produced on an industrial level—coupled with necessary greenhouse gas emission reductions—this could help stall climate change.
Professor Ian Power of Trent University in Ontario, Canada, who is heading the project, shared the latest discovery with Phys Org.
"Our work shows two things. Firstly, we have explained how and how fast magnesite forms naturally. This is a process which takes hundreds to thousands of years in nature at Earth's surface."
"The second thing we have done is to demonstrate a pathway which speeds this process up dramatically."
Reusable polystyrene microspheres were used in the research as a "catalyst" to accelerate the formation of magnesite inexpensively and at room temperature within 72 days.
"Using microspheres means that we were able to speed up magnesite formation by orders of magnitude. This process takes place at room temperature, meaning that magnesite production is extremely energy efficient."
Power added that their findings are still in the experimental phase and further tests are required.
But, he remains optimistic.
"For now, we recognise that this is an experimental process, and will need to be scaled up before we can be sure that magnesite can be used in carbon sequestration (taking CO2 from the atmosphere and permanently storing it as magnesite)."
"This depends on several variables, including the price of carbon and the refinement of the sequestration technology, but we now know that the science makes it do-able"
Giphy
To demonstrate the chemical reaction of how carbon dioxide is siphoned from the atmosphere into the magnesite, Forbes provided a breakdown of the formula.
CO2+ H2O→H2CO3→ H++ HCO3− then Mg+2+HCO3− →MgCO3+H+
According to the equation above, carbon dioxide is injected into water,
"which is then dissociated to form carbonic acid."
"From there, elemental magnesium combines with the carbonic acid to form magnesite (MgCO3)."
Forbes also noted that magnesite has the potential of pulling half its weight in carbon dioxide from the atmosphere.
"Estimates put our current CO2 emissions at about 40 billion tons per year."
"That would mean to remove the equivalent amount of carbon emitted per year solely through magnesite formation, 80 billion tons would have to be produced per year."
"It becomes increasingly apparent that this cannot be the only lever we pull in mitigating climate change."

But this breakthrough still sounds impressive even if it can't be the only solution.
Hopefully, there won't be any obstacles to prevent this from happening.










 @AngelaPaxtonTX/X
@AngelaPaxtonTX/X @EugeneWong3188/X
@EugeneWong3188/X @ImKnotTheOne/X
@ImKnotTheOne/X


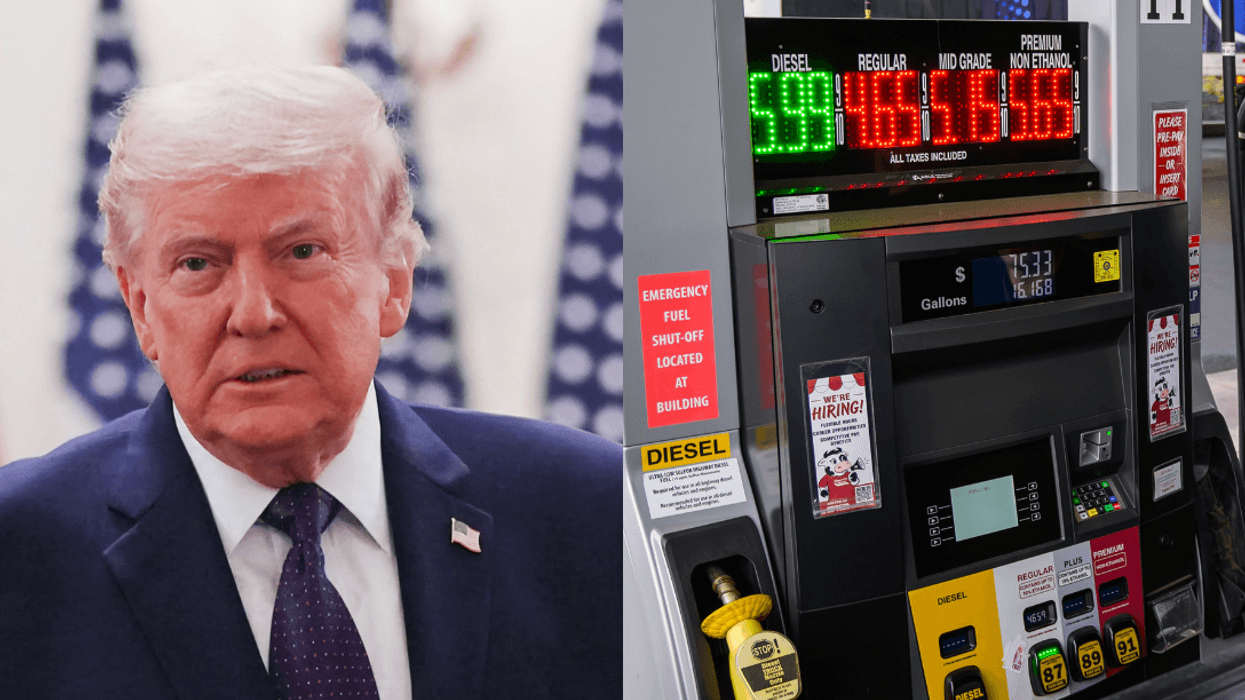


 @drewfromtv/Threads
@drewfromtv/Threads
 @WhiteHouse/X
@WhiteHouse/X

 @cleanpuppy/TikTok
@cleanpuppy/TikTok @itsjasminew/TikTok
@itsjasminew/TikTok @ashleybenson/TikTok
@ashleybenson/TikTok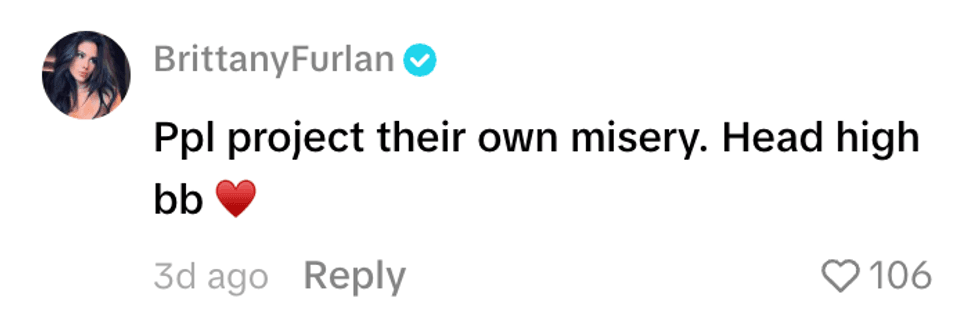 @brittanyfurlan/TikTok
@brittanyfurlan/TikTok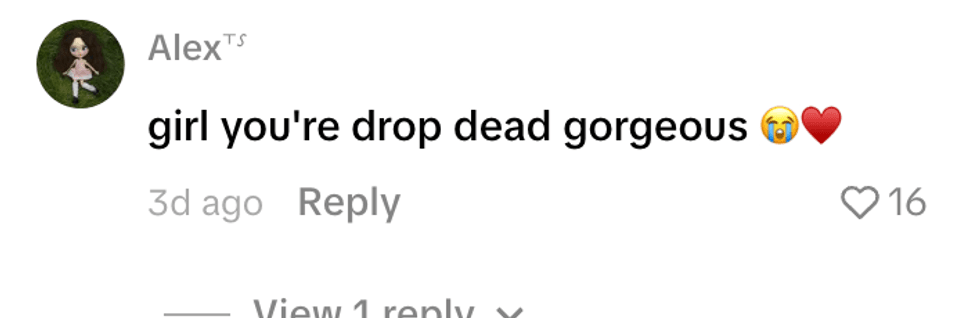 @alex_050873/TikTok
@alex_050873/TikTok @fanksrachel/TikTok
@fanksrachel/TikTok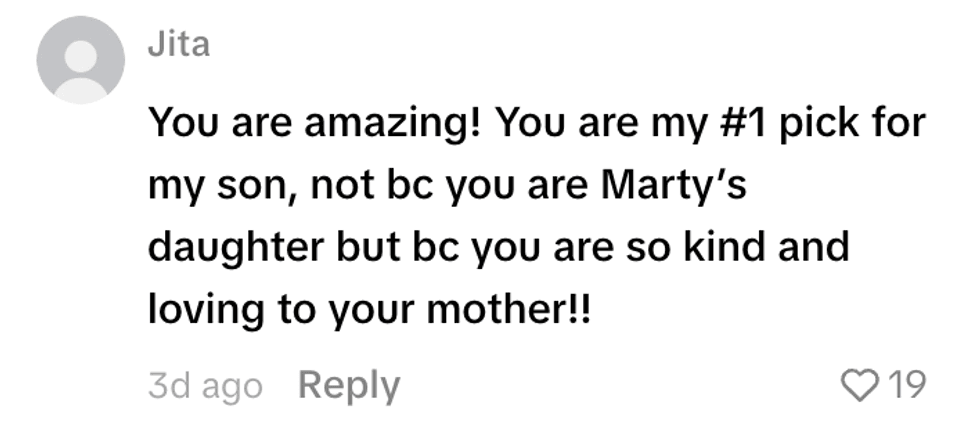 @aioop6/TikTok
@aioop6/TikTok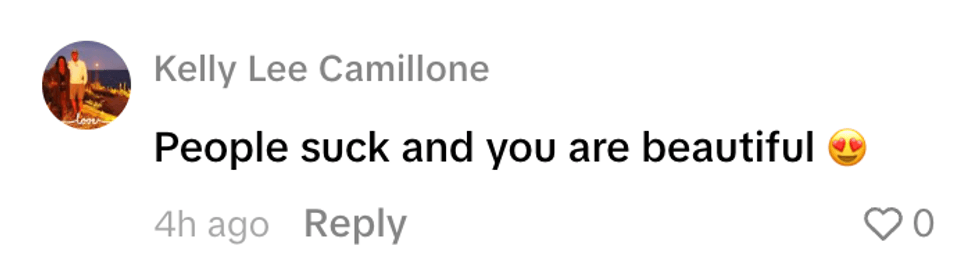 @kellyleecamillone/TikTok
@kellyleecamillone/TikTok @katieschuyler/TikTok
@katieschuyler/TikTok @nfltg_the_barbarian/TikTok
@nfltg_the_barbarian/TikTok @pohv_0/TikTok
@pohv_0/TikTok