For the first time, a federal advisory committee has recommended that the Food and Drug Association (FDA) vote to approve a prescription cannabidiol medicine. The medication, known as Epidiolex, is a form of cannabidiol (CBD), which is a cannabis derivative. According to a report by NBC News, approval of the medication would be limited to treating seizures caused by Lennox-Gastaut syndrome and Dravet syndrome in patients aged 2 and older. Although the FDA has indicated that it would vote in favor or approval, the medication has only received such a recommendation from the aforementioned federal advisory committee after a unanimous vote. The FDA is expected to make a final decision by June.
As previously reported in December, Dravet syndrome has already been treated in legal states by cannabis-oil derivatives. A study in the New England Journal of Medicine indicated that out of 120 children, half experienced reduction of the frequency of seizures when treated with medicinal cannabis-based products.
“Both Lennox-Gastaut syndrome and Dravet syndrome are rare, severe, refractory epilepsy syndromes with onset in early childhood,” the FDA said, in documents provided to the advisory committee.
“It’s not clear how or why CBD works, but studies show it does. The results from these three studies provide substantial evidence of the effectiveness of CBD for the treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome. In general, the risks associated with CBD treatment appear acceptable, particularly given the findings of clinical efficacy in LGS and DS, which are serious, debilitating, and life-threatening disorders.”
Historically, any form of CBD has been considered a Schedule 1 controlled substance. Schedule 1 medications are defined by the DEA as having no verifiable medical benefit. The pending approval of a cannabis medication demonstrates a notable shift in established cannabis policy.
“Epidiolex represents hope for the many individuals living with intractable seizures and rare epilepsies, who every day face incredible challenges and disabling seizures, and live with the continual risk of serious injury and death,” said Philip Gattone, president and CEO of the Epilepsy Foundation.
The internet currently contains thousands of CBD products. However, these products have not received FDA approval and their effectiveness as medications have not been scientifically substantiated. Previously, the FDA has approved a few other marijuana-based products, including Marinol, which is a THC-based medication used to treat severe weight loss in AIDS patients.
"It's very important to highlight that the drug used in this study, cannabidiol, was derived from cannabis plants, and purified to 99 percent purity," Devinsky told NBC News. "This is not something you can get from a dispensary today in the United States. It's not something that you can get by smoking marijuana. It is a very specific pharmaceutical grade product derived from marijuana, but quite different than what is available anywhere else right now in the United States."
News of the pending approval was met with widespread approval on various social media platform, with many users sharing the potentially historic decision:
[embed]
[/embed]CBD is different from other forms of marijuana, in that it does not contain THC, a necessary component needed to produce psychological effects and the typical “high” associated with marijuana.
Currently, 29 states have legalized medical marijuana, as the public narrative surrounding the medical benefits of marijuana continues to change.







 Roberto Schmidt/AFP via Getty Images
Roberto Schmidt/AFP via Getty Images





 u/pizzaratsfriend/Reddit
u/pizzaratsfriend/Reddit u/Flat_Valuable650/Reddit
u/Flat_Valuable650/Reddit u/ReadyCauliflower8/Reddit
u/ReadyCauliflower8/Reddit u/RealBettyWhite69/Reddit
u/RealBettyWhite69/Reddit u/invisibleshadowalker/Reddit
u/invisibleshadowalker/Reddit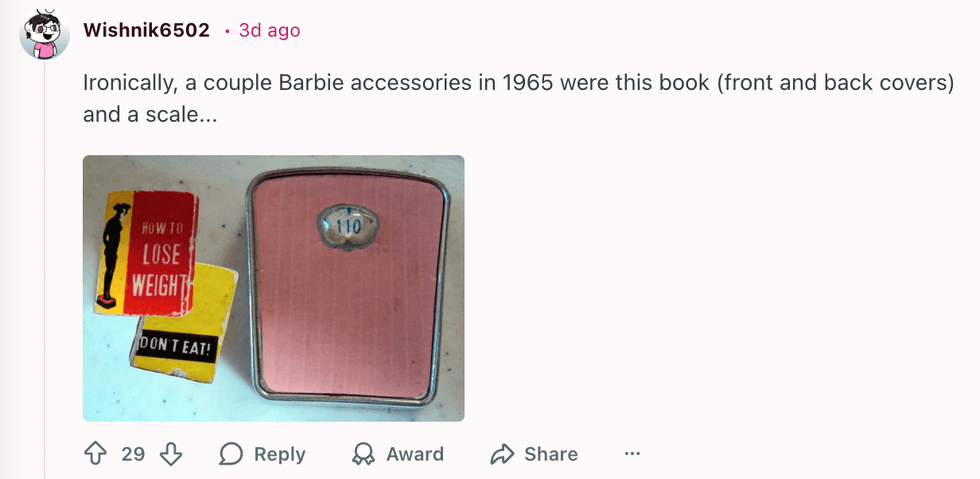 u/Wishnik6502/Reddit
u/Wishnik6502/Reddit u/kateastrophic/Reddit
u/kateastrophic/Reddit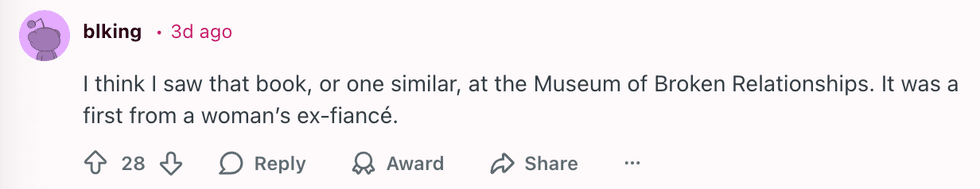 u/blking/Reddit
u/blking/Reddit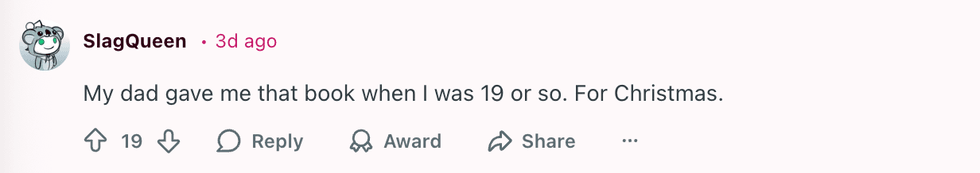 u/SlagQueen/Reddit
u/SlagQueen/Reddit u/geezeslice333/Reddit
u/geezeslice333/Reddit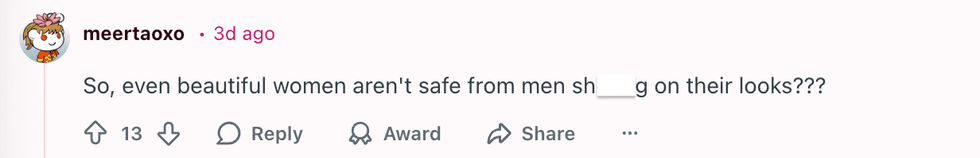 u/meertaoxo/Reddit
u/meertaoxo/Reddit u/crystal_clear24/Reddit
u/crystal_clear24/Reddit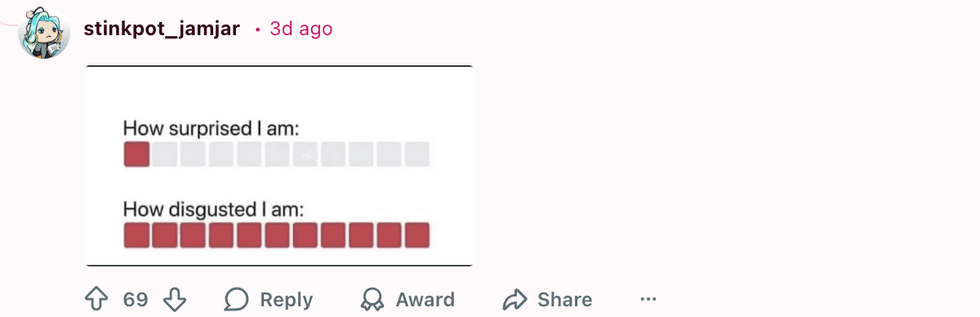 u/stinkpot_jamjar/Reddit
u/stinkpot_jamjar/Reddit
 u/Bulgingpants/Reddit
u/Bulgingpants/Reddit
 @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok @hackedliving/TikTok
@hackedliving/TikTok
 @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram @vanderjames/Instagram
@vanderjames/Instagram