Geologists have discovered a new type of water ice crystal inside diamonds brought to the surface from Earth's mantle.
The diamonds originated in the transition zone between the mantle and the outer core, roughly 410-660 kilometers below our planet's surface. The discovery of the ice, known as Ice-VII, is the first confirmation that Ice-VII occurs naturally on Earth. Previously, it had only shown up in laboratory experiments.
A new study published in Science outlines the findings, which are giving scientists new insights into the environment in an otherwise inaccessible part of our planet. "The transition zone is a region where the stable minerals have high water storage capacity," lead researcher Oliver Tauschner of the University of Nevada writes in the study. "The inclusions suggest that local aqueous pockets form at the transition zone boundary owing to the release of chemically bound water as rock cycles in and out of this region."
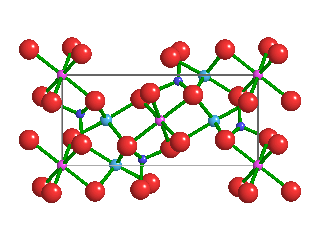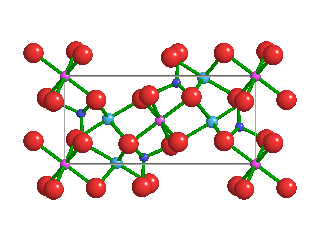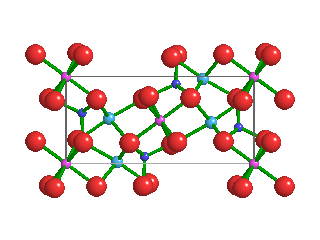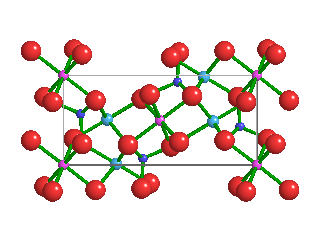
Like the diamonds in which it was found, Ice-VII is formed under extremely high pressures. Ice-VII is 1.5 times as dense as typical water ice, which scientists refer to as Ice-I. In Ice-I, water molecules form hexagonal crystalline patters. Because water molecules in Ice-VII form a cubic shape, however, they are very stable. As pressures increase, water ice transitions from one phase to another; Ice-I into Ice-II, Ice-II into Ice-III, and so on.
Pressures and temperatures inside Earth's mantle are enormous—as primordial diamonds are pushed upward through the mantle, they capture tiny bubbles of high-pressure water molecules. Ice-VII can't form directly inside the mantle, because the temperatures are too high. But when water molecules become trapped inside diamonds, the hardest naturally-occurring mineral on Earth, and those diamonds quickly rise toward Earth's crust, temperatures cool while immense pressures are maintained. This is what allows Ice-VII to crystallize inside diamonds.
"Usually the extremely deep minerals that come up to the surface are not stable once they experience low pressures," said Caltech geologist George Rossman, who worked on the study. "They crack and whatever inclusions they had in them are lost. But if a diamond comes up fast enough, it doesn't change."
Many scientific breakthroughs are made by accident, and the discovery of Ice-VII is no exception. "Water in diamonds is not unknown, but finding this very high pressure form of water ice intact, that was really fortuitous," Rossman said. "That's what you call discovery."
Though previously unknown to occur naturally on Earth, Ice-VII is believed to be present in other places in our solar system, such as on the Moon, Europa, and Enceladus. Since Ice-VII is very stable under high pressures and cold temperatures, these icy bodies may be the perfect locations for an abundance of Ice-VII to exist.










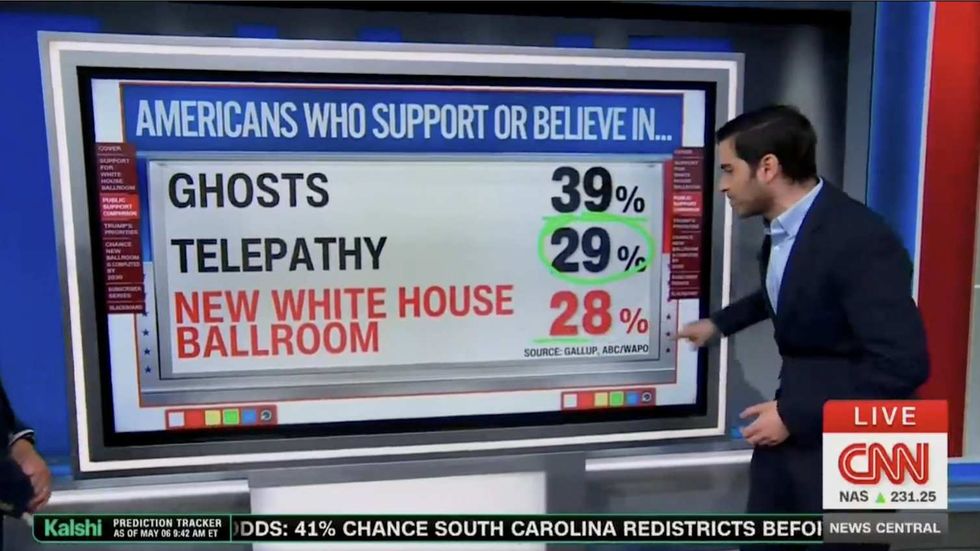





 @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
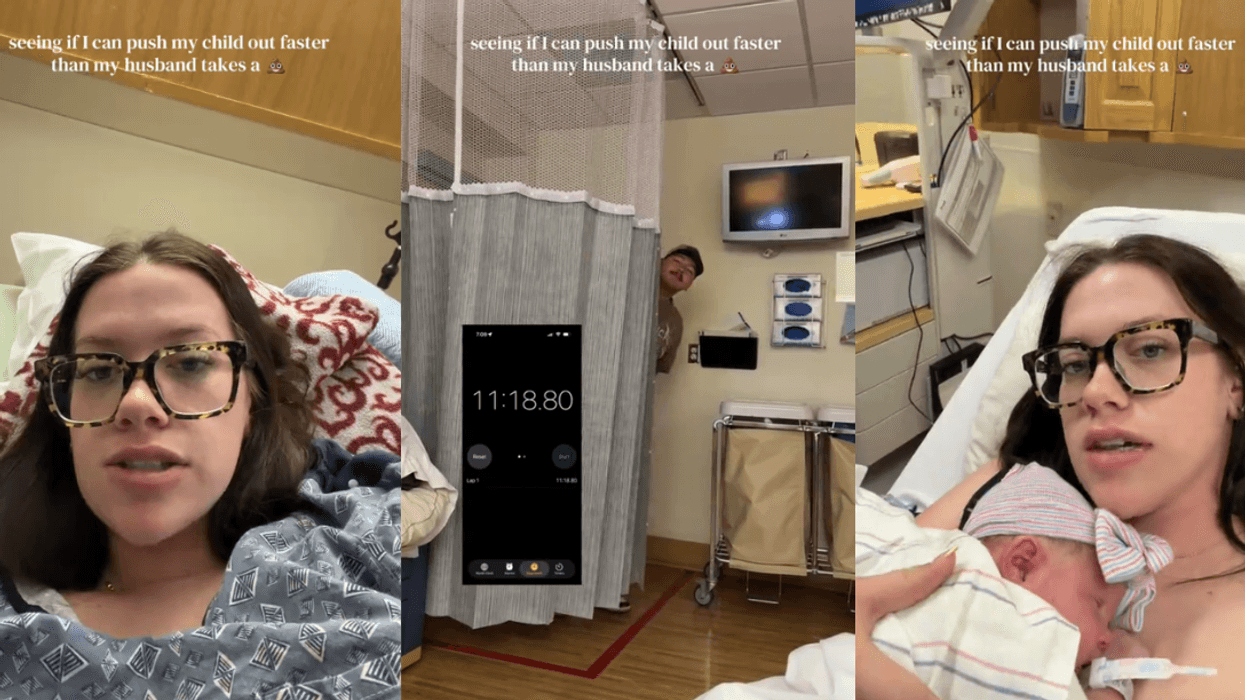
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok @devynnehaddoxx/TikTok
@devynnehaddoxx/TikTok

 Cover Images
Cover Images
