Donald Trump Jr. was swiftly criticized after he attempted to blame a nationwide infant formula shortage on President Joe Biden, saying the shortage is a reflection of "Biden's America."
"Let that sink in," said Trump Jr., who is the latest Republican to comment as the United States grapples with an infant formula shortage due to supply chain issues and product recalls.
Trump Jr. later criticized the Biden administration for sending $40 billion in aid to Ukraine as it continues to fend off Russian President Vladimir Putin's forces in a war that is now in its third month.
Trump Jr. suggested that the decision to send aid to Ukraine is a sign that the Biden administration is less concerned with domestic issues, saying that Congress should "put America First for a change."
The nationwide infant formula shortage has received renewed attention in recent days as the Food and Drug Administration (FDA) works with Abbott Nutrition, a company that had to recently recall its product, to safely resume production.
As of early April, "seven states reported that between 40% and 50% of baby formula products were out of stock," according to a CNN report.
Experts say this is the worst shortage the United States has experienced in decades, amplified by Abbott Nutrition's prolonged shutdown as well as general supply chain issues that have made securing formula ingredients and packaging increasingly difficult.
This week, New York Attorney General Letitia James said that her office is "closely monitoring reports of a baby formula shortage impacting families across our nation" and said that the state government "won't hesitate to take action against anyone who price gouges baby formula."
Trump Jr.'s statements were harshly criticized and many pointed out that recent Republican support for the dismantling of abortion rights could no doubt complicate the infant formula shortage further.
Abbott Nutrition has said that it hopes that its Sturgis, Michigan plant could be running within two weeks and that their products could be back on shelves within six to eight weeks, pending FDA approval. The plant will have improved systems and protocols in place to guard against future contamination.
The company said it "would begin production of EleCare, Alimentum and metabolic formulas first and then begin production of Similac and other formulas" with the FDA's go-ahead.















 @johngriff9ne/Threads
@johngriff9ne/Threads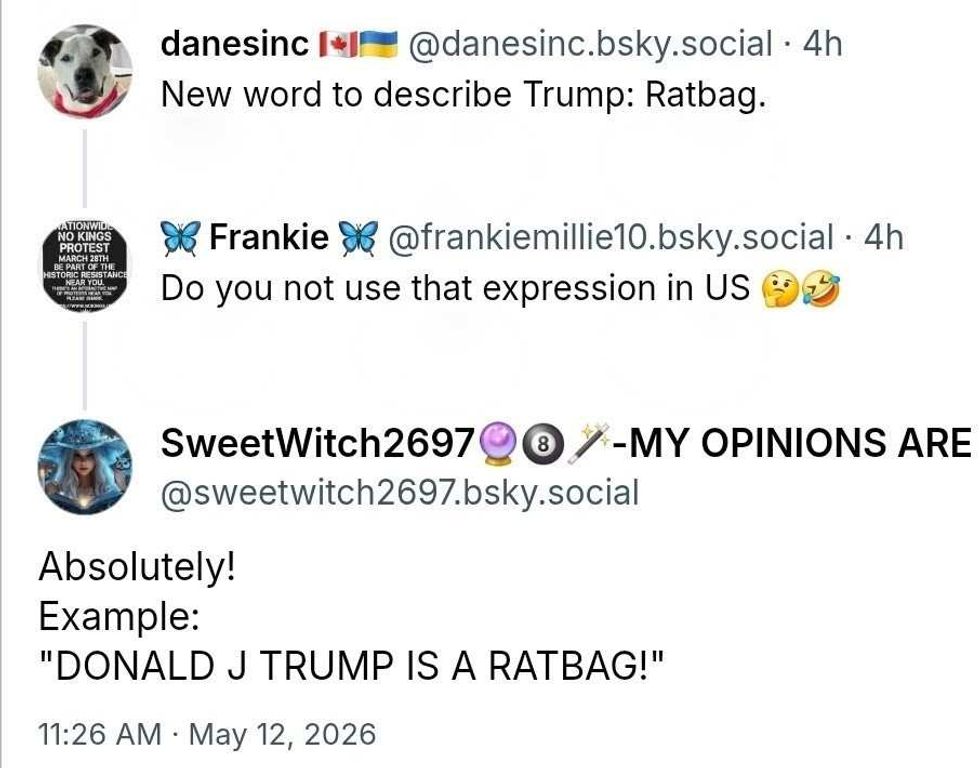 @danesinc; @frankiemillie10; @SweetWitch2697/Bluesky
@danesinc; @frankiemillie10; @SweetWitch2697/Bluesky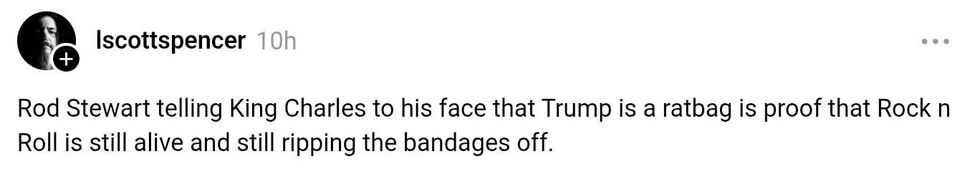 @lscottspencer/Threads
@lscottspencer/Threads @gregproops/Bluesky
@gregproops/Bluesky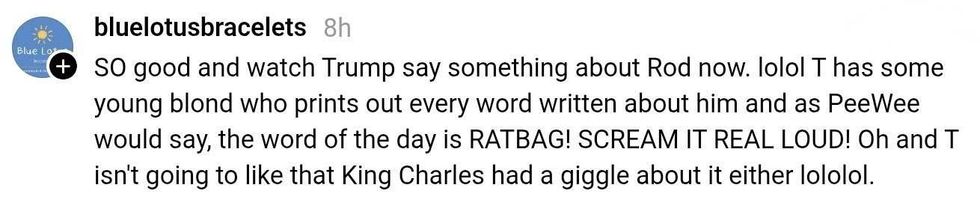 @bluelotusbracelets/Threads
@bluelotusbracelets/Threads @sorry-iamcanadian/Bluesky
@sorry-iamcanadian/Bluesky @fubarrockchick/Bluesky
@fubarrockchick/Bluesky @accidentaldomesticgoddess/Threads
@accidentaldomesticgoddess/Threads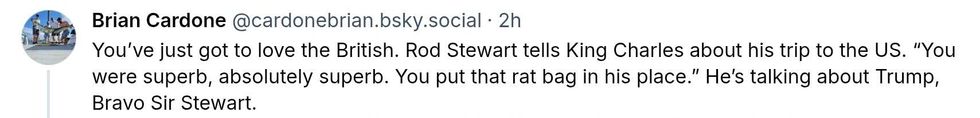 @cardonebrian/Bluesky
@cardonebrian/Bluesky @DividerDecider/Bluesky
@DividerDecider/Bluesky @samjcaruso/Threads
@samjcaruso/Threads @kathyfinch311/Threads
@kathyfinch311/Threads

 @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram @dcoulier/Instagram
@dcoulier/Instagram